Latest News, 12/18/21: Beacon Medical Germany started the distribution with medical cannabis flowers in Germany in cooperation with Medical Pharma Resource.
Medical cannabis
Beacon Medical has all necessary permits to import and distribute medical cannabis, the wholesale permit according to § 52a AMG, GMP / GDP certificate, narcotics permit according to §3 BtMG, import permit according to § 72 AMG. We import and distribute medical cannabis with the highest reliability. We offer doctors, pharmacists and patients quick orientation, forward-looking therapy solutions and first-class service.
We are Beacon Medical Germany.
VALUABLE INFORMATION
What is medical cannabis?
Medical cannabis is a basically versatile possible therapy option. The reason for this is the different concentration as well as the variety of its different…
Read moreManufacture and use of medical cannabis
Why is the production of medical cannabis so expensive? And how can it be taken? You can find the answers here. Small branches, the so-called…
Read moreTherapy option: Medical cannabis
For which diseases can medical cannabis be prescribed? And in which situations do the health insurance companies cover the costs? A brief summary of the…
Read more
Beacon Medical Germany GmbH is a young pharmaceutical enterprise based in Potsdam, near Berlin.
We are the German subsidiary of the Canadian VIVO Cannabis Inc. The market-listed VIVO Cannabis Inc. is a company with subsidiaries engaged in the cultivation, production and marketing of medical cannabis.
In cooperation with our experienced Canadian sister company ABcann Medicinals Inc., a producer specialising in medical cannabis since 2014, we realise the distribution and marketing of medical cannabis.
Harnessing the potential of medical cannabis
As a reliable partner, we support doctors, pharmacists and patients in treatment with medical cannabis. We rely on the quality of our products and services, with the aim of enabling patients to benefit as much as possible from this innovative therapeutic option and improving their quality of life.
Quality
Our standardised production with optimal growth conditions guarantees medical cannabis of consistent, certified quality.
Service
Beacon Medical Germany is a competent partner for doctors, pharmacists and patients in treatment with medical cannabis.
We believe in the future of medical cannabis as a valuable therapeutic option in medicine and want to actively shape this development.

Career
LOOKING FOR A NEW CHALLENGE?
You are welcome to actively participate in the growth of our company and to make your plans for the future a reality with us.
Apply to Beacon Medical Germany!
Beacon Medical has all necessary permits to import and distribute medical cannabis, the wholesale permit according to § 52a AMG, GMP / GDP certificate, narcotics permit according to §3 BtMG, import permit according to § 72 AMG. We import and distribute medical cannabis with the highest reliability. We offer doctors, pharmacists and patients quick orientation, forward-looking therapy solutions and first-class service.
Medical cannabis is obtained from the hemp plant Cannabis sativa L. The flowers in particular are rich in natural substances such as cannabinoids. The two most important among them are THC (Δ9-tetrahydrocannabinol) and CBD (cannabidiol). Medical cannabis is cultivated, harvested, processed and analysed under strictly controlled conditions so that high quality can be guaranteed for the patient.
Medicinal cannabis is used primarily to treat pain, and also for the treatment of multiple sclerosis and epilepsy. It may be prescribed if no approved treatment alternatives are available, previous treatments have not been sufficiently effective or have not been tolerated.
All doctors who can issue prescriptions for narcotics are allowed to prescribe medical cannabis. Check with official associations or patient groups and discuss treatment options with your doctor in person.
Medical cannabis is classified as a prescription narcotic. Only professionals such as doctors or pharmacists may receive detailed information on such products from the manufacturers. Companies selling medical cannabis are not allowed to give any information directly to the patient outside of these specialist circles. To discuss whether cannabis therapy is an option for you, you should contact your doctor. He or she can inform you about the possibilities of cannabis therapy and, if necessary, help you find a suitable product in a suitable dosage for you.
The statutory health insurance funds cover the costs for treatment with cannabis for serious illnesses if no alternative therapy is available, previous therapies were not sufficiently effective or were not tolerated. There are forms that you can fill out together with your doctor. The health insurance company will decide on this application for reimbursement of costs within three weeks. If the medical service of the health insurance companies is involved, the decision can take five weeks.
If the application is rejected, the patient can lodge an appeal with their doctor within one month. A private prescription is also possible. However, the costs must then be borne by the patient themselves.
The deadlines for palliative care situations are different: In the final stage of a serious illness, the health insurance funds must announce their decision on an application for reimbursement after three days. If this deadline is exceeded, the application for reimbursement is considered approved.
According to current legislation, medical cannabis is only reimbursed by health insurance funds if there are no alternative therapies available for a serious disease, if previous therapies have not been sufficiently effective or if they have not been tolerated. The legislature has established this for medical cannabis because there are fewer clinical trials compared to other treatment options. Such studies are important in medicine for assessing the efficacy and risks of a product.
However, there are many different medical cannabis products with different levels of THC and CBD. Of course, studies are not available for every product for every disease. Therefore, the doctor prefers to prescribe drugs with a better study situation.
Medical cannabis is produced under controlled conditions and guarantees high quality to the user. Cannabis not approved for medical purposes does not meet these requirements and its purchase is illegal in Germany. A reimbursement from the health insurance companies is then, of course, also excluded.
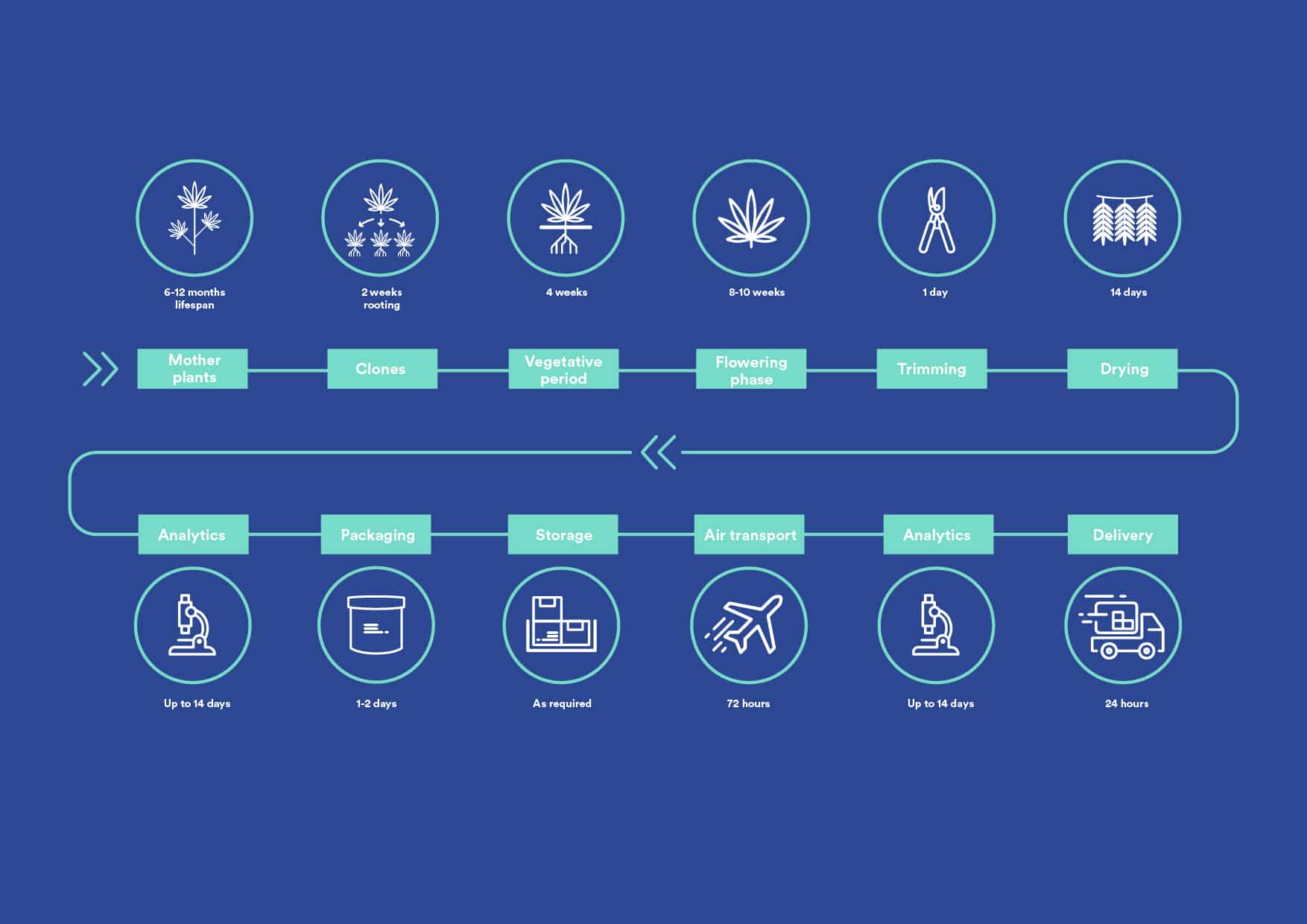
Clones are taken from carefully selected cannabis mother plants. These clones are then planted under controlled conditions and grown until they bloom. The harvested cannabis flowers can be used to produce medical cannabis extracts or they can be dried. The dried whole cannabis flowers are packaged and thoroughly analysed before being released for sale.
Medical cannabis also has an addictive potential, which is why it belongs to the group of narcotics like other highly effective pain killers. When prescribing medical cannabis, your doctor will point out the potential for addiction and try to keep the risk as low as possible.
VIVO is a family of premium brands and businesses, building a world-class cannabis company through the acquisition and development of specialty brands.